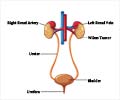
New treatment approach increases four-year overall survival to 12% for patients with stage IV Wilms tumor with lung metastases.
‘New treatment approach will reduce the need for radiation and the long-term risks associated with it.’





The standard treatment for Wilms tumor with lung metastases in North America is to give "3-drug" chemotherapy (vincristine/dactinomycin/doxorubicin) and lung RT. The AREN0533 study assessed whether lung RT can be avoided for patients with complete lung nodule response after six weeks of chemotherapy. Conversely, the study assessed the benefit of adding two additional chemotherapy agents, cyclophosphamide and etoposide, to the treatment regimen for patients with incomplete lung nodule response, a treatment strategy that was associated with inferior outcomes in previous studies. AREN0533 was also the first study in Wilms tumor to modify treatment based on an adverse biological prognostic factor, loss of heterozygosity (LOH) at chromosomes 1p and 16q. The study showed: • The approach to therapy based on lung nodule response and LOH at chromosomes 1p and 16q resulted in a 4-year overall survival rate of 96 percent, compared to 84 percent on the predecessor study.
• About 40 percent of patients with Wilms tumor and lung metastases can be spared initial upfront lung radiation and still have outstanding survival. This will decrease the long-term risk of heart toxicity and breast cancer.
• Patients with incomplete lung nodule response after six weeks of therapy with cyclophosphamide and etoposide had significantly better 4-year event-free survival: 89 percent compared with 75 percent that was expected based on historical data.
• Intensification of therapy for patients with LOH at 1p and 16q was highly effective: 4-year event-free survival rate improved from 66 percent on the previous study to 100 percent.
Advertisement
Advertisement