A new study has shown that butylphthalide, a medicine derived from celery seed can help treatment outcomes of stroke patients.
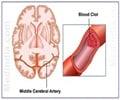
- A stroke occurs when something blocks blood supply to part of the brain or when a blood vessel in the brain bursts, which can cause parts of the brain to become damaged or even die
- A recent study suggests that 90 days after a clot-caused stroke, people who received a celery seed-derived medication in addition to prompt treatment for the clot had less severe neurological symptoms and better functioning in comparison to those who received clot treatment and a placebo medication
- The celery seed-derived medication, called butylphthalide, may help protect brain tissue from damage when the supply of oxygenated blood is cut off
Butylphthalide is approved and available for use in China, where the study was done. However, the medication hasn’t been approved for use by the FDA.
Effect of Butylphthalide on Stroke
“This is the first trial to show the benefit of using a medication that protects the brain from damage caused by a lack of oxygen to brain tissue. The medication was given to patients with acute ischemic stroke who were also receiving treatment to restore blood flow to the brain,” says co-author Baixue Jia, MD, a doctor of interventional neuroradiology at the Beijing Tiantan Hospital and the China National Clinical Research Center for Neurological Diseases in Beijing.“Patients who received butylphthalide had less severe neurological symptoms and a better living status at 90 days post-stroke, compared to those who received the placebo,” Jia says. “If the results are confirmed in other trials, this may lead to more options to treat strokes caused by clots.”
The findings were presented at the American Stroke Association’s International Stroke Conference.
Previous animal research in China has indicated that butylphthalide seems to have the potential to protect the brain from harm caused by a clot in the brain. Human testing has now started.
90-Day Outcomes of Butylphthalide
Researchers studied whether treatment with butylphthalide could improve 90-day outcomes in adults who also received standard clot treatment, such as an IV-given clot-busting medication called tissue plasminogen activator, or tPA, mechanical clot removal, or both.The patients were treated at one of 59 medical centers in China between 2018 and 2022. Along with the initial clot treatment chosen by their doctor, they were randomly selected to receive either butylphthalide or a ‘look-alike’ placebo, which were given as daily shots for the first 14 days and then as oral capsules for 76 days. Neither the patients nor the researchers knew how the people in the study were assigned.
The researchers defined the outcomes as ‘favorable’ if a patient fell into one of these categories at 90 days after the stroke: an initially mild to moderate stroke and no symptoms after treatment; an initially moderate to serious stroke and no symptoms or mild symptoms that didn’t stop them from doing daily activities after treatment; or an initially serious to severe stroke and no symptoms or a slight disability that impaired some activities after treatment.
Among the 1,216 patients, 607 took butylphthalide, and 609 took the placebo. The average age was 66 years, and 68% of the study participants were men.
Favorable Outcome of Butylphthalide
Overall, people in the butylphthalide group were 70% more likely to have a favorable 90-day outcome, as compared with the placebo group.Butylphthalide also improved function among all the patients, including those who initially received tPA, those who received mechanical clot removal, and those who received both.
Other outcomes such as recurrent stroke and brain bleeding weren’t significantly different between the two groups.
The authors noted that more studies are needed. Researchers are still not able to completely understand how butylphthalide works in the body or how stroke patients may respond if they receive treatments other than tPA or mechanical clot removal.
While these are interesting results, this is only one relatively small study on a fairly select population in China.
There is, however, a need to be cautious about directly associating butylphthalide with celery seeds or celery seed extract supplements.
“While this derivative that seems to be effective comes from celery seeds, it’s not the same as eating celery seeds. Stroke survivors should always consult with their neurologist or health care professional regarding diet after a stroke.”
Reference:
- Novel celery seed-derived medicine given after clot treatment may improve stroke outcomes - (https://newsroom.heart.org/news/novel-celery-seed-derived-medicine-given-after-clot-treatment-may-improve-stroke-outcomes?preview=5164.)
Source-Medindia