Lower dose of monkeypox vaccine is found to be safe with equal six-week antibody response, promising for vaccination strategies.

Safety and Immunogenicity of Modified Vaccinia Ankara-Bavarian Nordic Smallpox Vaccine in Vaccinia-Naive and Experienced Human Immunodeficiency Virus-Infected Individuals: An Open-Label, Controlled Clinical Phase II Trial
Go to source). The Mpox virus has been present in west, central and east Africa for decades, with the first human case identified in 1970. In May 2022, a global Mpox outbreak caused by the clade IIb strain of the virus provided the first epidemiologic evidence of community Mpox transmission outside of historically affected countries.
‘Lower dose #monkeypox vaccine ensures safety while maintaining an equivalent six-week antibody response, offering a promising alternative for vaccination strategies. #mpox #vaccine’





Dose-Sparing Strategies for MVA-BN (JYNNEOS) Vaccine
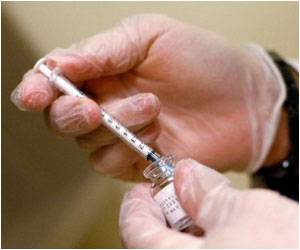
The Modified Vaccinia Ankara-Bavarian Nordic (MVA-BN, sold as JYNNEOS) vaccine was made available to help contain the outbreak in the United States. The National Institutes of Health’s (NIH) National Institute of Allergy and Infectious Diseases (NIAID) sponsored a study of dose-sparing strategies to extend the limited vaccine supply.The mid-stage study enrolled 225 adults aged 18 to 50 years in the United States who had not previously been vaccinated against Mpox or smallpox. Participants were randomized to receive either the standard Food and Drug Administration-approved MVA-BN regimen, a regimen containing one-fifth of the standard dose, or one with one-tenth of the standard dose. The standard dose was injected under the skin (subcutaneously), while the dose-sparing regimens were injected between layers of the skin (intradermally). Participants in all study arms received two injections 28 days apart and were monitored for safety and immune response.
Two weeks after the second dose (study day 43), participants who received one-fifth of the standard dose had antibody levels equivalent to those of participants receiving the standard MVA-BN regimen, based on predefined criteria. By day 57, participants who received one-fifth of the standard dose had lower antibody levels than those in the standard regimen arm; the clinical significance of this difference is unknown. Participants who received one-tenth of the standard dose had inferior antibody levels at all measurements. The most reported adverse events were mild, local injection-site reactions. Adverse events were similar across all arms of the trial, and no serious adverse events related to the vaccine were reported.
The authors note that because there are no defined correlates of protection against Mpox —immune processes confirmed to prevent disease—these findings cannot predict the efficacy of dose-sparing regimens with certainty. Real-world data from the Centers for Disease Control and Prevention and others have shown similar vaccine effectiveness for the dose-sparing regimen given intradermally and the standard regimen given subcutaneously. A study of the standard MVA-BN regimen in adolescents is ongoing and will report findings later this year.
Reference:
- Safety and Immunogenicity of Modified Vaccinia Ankara-Bavarian Nordic Smallpox Vaccine in Vaccinia-Naive and Experienced Human Immunodeficiency Virus-Infected Individuals: An Open-Label, Controlled Clinical Phase II Trial - (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4567089/)
Source-Eurekalert