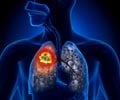
In patients with locally advanced non-small cell lung cancer (NSCLC), the addition of drugs oleclumab or monalizumab to durvalumab improved progression-free survival.
“Our results from the COAST trial are very encouraging as the data suggest these novel immunotherapy drug combinations could improve clinical outcomes for patients with advanced NSCLC,” said Roy S. Herbst, MD, PhD, Chief of Medical Oncology at Yale Cancer Center and Smilow Cancer Hospital and senior author of the study.
“One in four patients with NSCLC are diagnosed where the majority of their tumors can’t be removed with surgery, however advances in chemotherapy and radiation therapy have proven effective. We are always striving to make new, more effective therapies post radiation to be available.”
COAST is an open label, randomized, phase 2 trial that studied at adjuvant therapy with durvalumab alone or in combination with oleclumab or monalizumab in patients with locally advanced, unresectable, stage III NSCLC. Durvalumab works with a patient’s immune system to interfere with the growth and spread of cancer cells in the body. Results show oleclumab, an anti-CD73 monoclonal antibody, or monalizumab, an anti-NKG2A monoclonal antibody, in combination with durvalumab improved progression-free survival.
After a follow-up of 11.5 months, the data revealed durvalumab in combination with oleclumab reduced the risk of disease progression or death by 56%, and in combination with monalizumab by 35%, when compared to durvalumab alone. The 10-month progression free survival rate was 64.8% for the durvalumab plus oleclumab combination and 72.7% for durvalumab plus monalizumab, versus 39.2% with durvalumab alone.
“These findings support further evaluation of these drug combinations,” said Herbst.
Advertisement