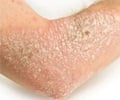
In a recent study it was found that certain biologic treatments to treat psoriasis do not increase risk for cardiovascular problems.
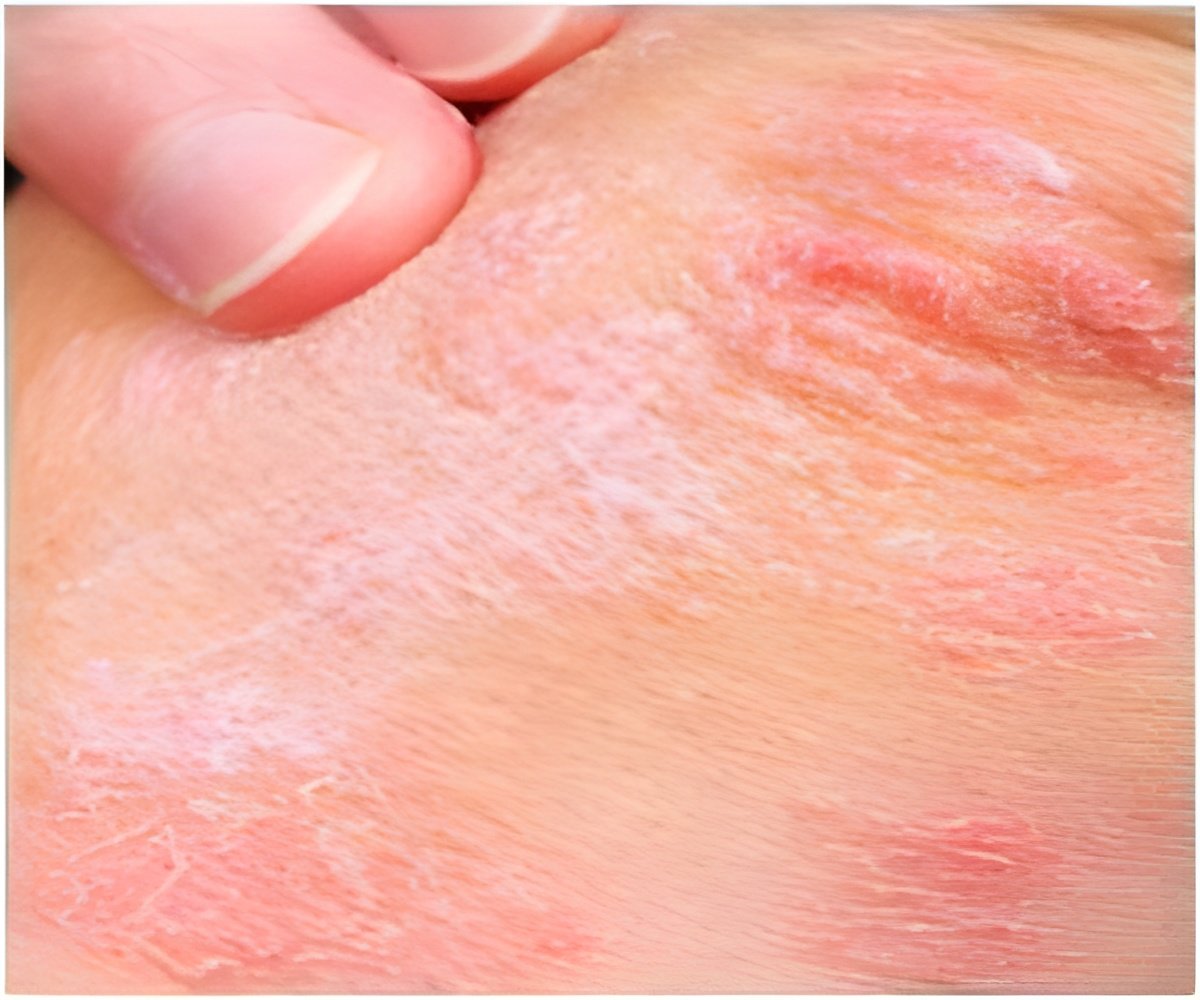
Caitriona Ryan, M.B., B.A.O., B.Ch., of the Baylor Research Institute, Dallas, and colleagues conducted a meta-analysis to evaluate a possible association between biologic therapies for chronic plaque psoriasis (CPP; the most common form of psoriasis, characterized by well-defined patches of red raised skin) and MACEs. The researchers indentified for inclusion in the analysis 22 randomized controlled trials of monotherapy studies (with safety outcome data for MACE) of anti-IL-12/23 agents (ustekinumab and briakinumab) and anti-TNF-α agents (adalimumab, etanercept and infliximab) in adults. The total number of participants in these studies was 10,183 patients. The primary outcome that was measured was a MACE during the placebo-controlled phase of treatment in patients receiving at least 1 dose of study agent or placebo.
During the placebo-controlled phases of the anti-IL-12/23 studies, 10 of the 3,179 patients treated with these therapies had a MACE compared with no events in the 1,474 patients treated with placebo. In studies of anti-TNF-α agents, 1 of the 3,858 patients receiving these agents had a MACE compared with 1 of the 1,812 treated with placebo.
"This meta-analysis did not show a significant increase in the risk of MACEs associated with the use of anti-lL-12/23 agents. Limitations of this study, however, prevent us from determining whether these drugs expose psoriasis patients to increased cardiovascular risk. Access to patient-level data for these studies was not granted by any of the study sponsors, which precluded the use of a more statistically robust time-to-event analysis. The small number of MACEs that occurred in placebo-controlled phases of these studies and the limited duration of the placebo-controlled phases reduce the power of this meta-analysis to detect a change in risk," the authors write.
The researchers add that this analysis highlights the inherent limitations of placebo-controlled, clinical trials to reliably interpret the significance of rare events given their current design. "Although RCTs are currently the criterion standard for measuring clinical efficacy in psoriasis therapies, these studies are designed to detect differences in the severity of psoriasis in response to therapy over short periods of treatment and are often underpowered and of too short duration to detect rare or long-term adverse events. Careful consideration of these issues is warranted to best serve patients in these studies and those who are treated once drugs are approved."
Source-Eurekalert