Stanford researchers have identified how a disease-associated protein TG2 gets inactivated, opening door to possible new treatments for celiac disease.

‘Blocking enzyme transglutaminase 2, or TG2 is a promising avenue for treating celiac disease patients without requiring lifelong changes to their diets.’





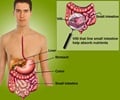
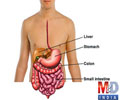
The biochemistry of how TG2 interacts with gluten and induces an immune response has been well studied, but more basic mysteries remain, for example how TG2 behaves in people without celiac disease. Chaitan Khosla, the professor at Stanford and director of Stanford Chemistry, Engineering & Medicine for Human Health who oversaw the new study, has conducted several studies showing that TG2 can be active or inactive, depending on the forming or breaking of a specific chemical bond, called a disulfide bond, between two amino acids in the enzyme. "(E)ven though there's a lot of transglutaminase 2 protein in the (small intestine), it's all inactive," Khosla said. "When it became clear that even though the protein was abundant, its activity was nonexistent in a healthy organ, the question became 'What turns the protein on, and then what turns the protein off?'" In 2011, Khosla's team identified the enzyme that activates TG2 by breaking its disulfide bond. In the new paper, the researchers performed experiments in cell cultures and found an enzyme that re-forms this bond, inactivating TG2. This enzyme, ERp57, is mainly known for helping fold proteins inside the cell. When it turns off TG2, it does so outside of cells, raising more questions about its functions in healthy people.
"Nobody really understands how (Erp57) gets outside the cell," Khosla said. "The general thinking is that it's exported from the cell in small quantities; this particular observation suggests that it actually does have a biological role outside the cell."
TG2 is now also the first protein known to have a reversible disulfide bond on/off switch of this type. "This is a very different kind of on-and-off chemistry than the kind that medicinal chemists would (typically) use," Khosla said.
Understanding this mechanism has led the team to investigate whether there are any FDA-approved drugs that could target the switch directly. Because previous studies have suggested that lack of TG2 doesn't seem to negatively affect the health of mice.
Advertisement